Thrombolex is committed to helping provide a new standard of care for the treatment of arterial and venous thromboembolic (A+VTE) conditions. Discover our innovative approach to treatment with the BASHIR™ Family of Endovascular Catheters.
An Elegant, Novel Solution
For Optimal Thrombus Resolution
The Problem
Treating A+VTE safely and effectively while minimizing risk, maximizing clot burden resolution, and producing consistent clinical outcomes is of critical importance.
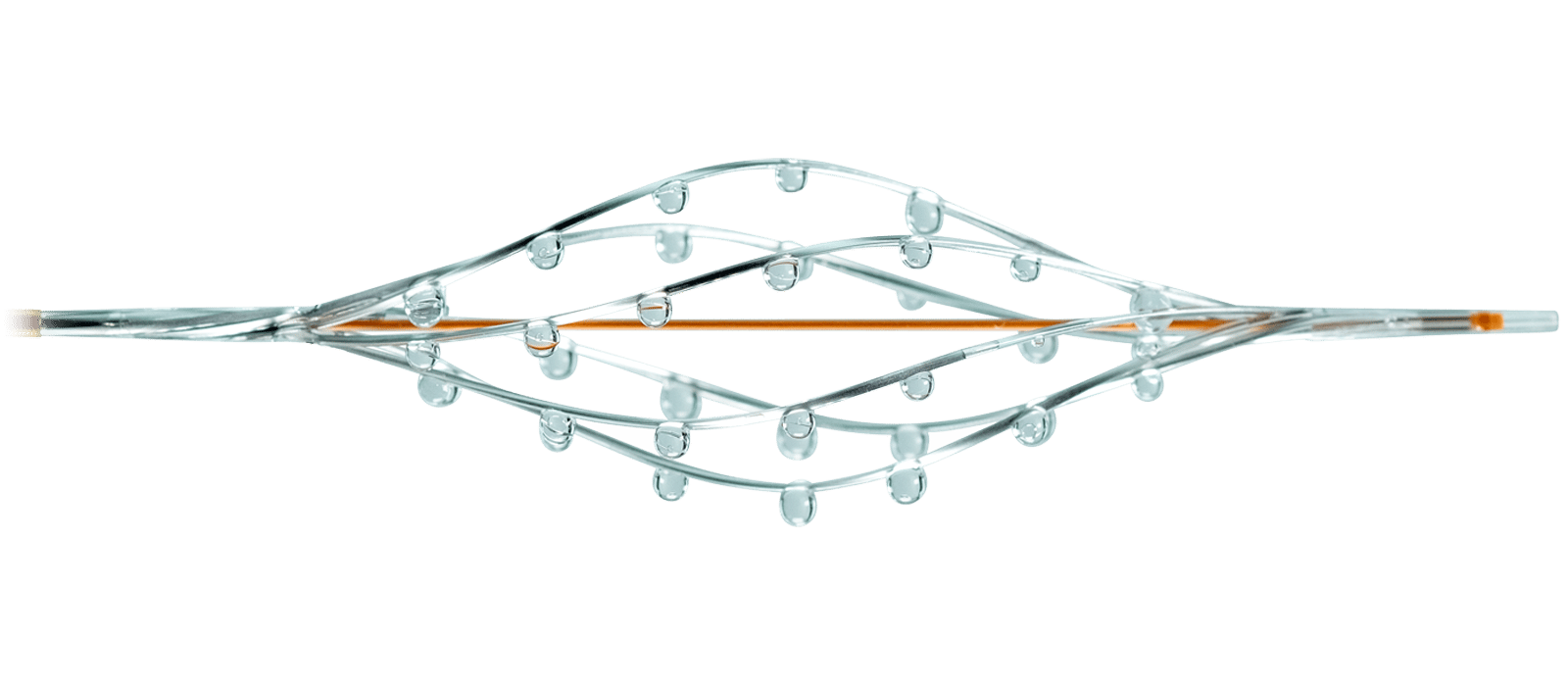
Our Solution
The unique hybrid mechanism of action of the BASHIR™ family of endovascular catheters allows the use of both mechanical and pharmacological treatment to immediately restore blood flow. This allows for the efficient and safe introduction of thrombolytics to produce positive clinical outcomes.
Product Resources
Product Overview • Product Details
Clinical Resources
Clinical Studies • Instructional Videos
Our Company
Our Mission • Our Leadership
Our Family of Catheters
For restoring blood flow in peripheral and pulmonary vessels
The catheter’s expandable infusion basket allows a controlled and selective infusion of physician-specified fluids.
Available in 7F and 8F sizes.
For restoring blood flow within shorter peripheral and pulmonary vessels
The short basket of the S-B catheter allows navigation and implementation in shorter, more tortuous vessels.
Available in 7F and 8F sizes.
For restoring blood flow in peripheral vessels with extensive thrombus burden
A family of catheters designed to deliver thrombolytics through the expandable infusion basket and along a segment of the catheter shaft.
–







Not all venous thrombus presents in the same way, and not all devices are built to address that variability.
The BASHIR® Plus Endovascular Device is designed for the treatment of venous thrombus with extensive clot burden, offering variable shaft infusion lengths - available in +10, +20, +30, and +40 configurations - to support mechanical fragmentation along with targeted uniform thrombolytic delivery across a range of anatomical presentations. The expandable infusion basket enables precise pharmaco-mechanical lysis without the need for capital equipment, making it straightforward to integrate into existing lab workflows.
Thrombolex.com #thrombolex ... See MoreSee Less
0 CommentsComment on Facebook
The evidence for single-session PML continues to grow.
Interim data from the first 50 patients in the RAPID-PE Study - a prospective, multicenter U.S. study evaluating on-the-table (OTT) pharmaco-mechanical lysis (PML) - was presented at TCT 2025. The analysis demonstrated that on-the-table PML using the BASHIR® Endovascular Device can be performed efficiently and without post-procedure infusion, offering the interventional community meaningful insight into what structured, targeted PE intervention looks like in practice.
As the study continues to enroll, the RAPID-PE Study reinforces the clinical case for single-session care in acute PE treatment.
Learn more at Thrombolex.com. #thrombolex ... See MoreSee Less
0 CommentsComment on Facebook
The results of the RESCUE-II Study have been published in JACC: Advances, and the data speaks for itself.
This peer-reviewed publication validates the role of the BASHIR® Endovascular Device as a clinically proven solution in the treatment of A&VTE, demonstrating significant reductions in RV/LV ratio and PA obstruction with no major bleeding events and no requirement for post-procedure infusion. All achieved in a single, on-the-table (OTT) pharmaco-mechanical lysis (PML) session.
In PE care, clinical confidence is earned through evidence. The RESCUE-II Study delivers exactly that.
Read the full publication in JACC: Advances at Thrombolex.com. #Thrombolex ... See MoreSee Less
0 CommentsComment on Facebook
Clinical confidence is built on evidence, and the RESCUE-II Study delivers.
In the RESCUE-II Study, patients treated with the BASHIR® Endovascular Device for acute pulmonary embolism demonstrated significant reductions in RV/LV ratio and PA obstruction, with no major bleeding events and no requirement for post-procedure infusion - all in a single session.
Learn more here: www.jacc.org/doi/10.1016/j.jacadv.2025.101789
Thrombolex.com #thrombolex ... See MoreSee Less
0 CommentsComment on Facebook
When treating pulmonary embolism, focus matters at every step.
The BASHIR® Endovascular Device is designed to support targeted thrombolytic delivery and mechanical clot fragmentation, helping clinicians maintain procedural consistency and clinical confidence throughout a single, on-the-table (OTT) pharmaco-mechanical lysis (PML) session.
Thrombolex.com #thrombolex ... See MoreSee Less
0 CommentsComment on Facebook
Evidence-based medicine is not a preference in pulmonary embolism treatment. It is a responsibility. Every clinical choice - the approach, the therapy, the protocol - shapes what happens next for the patient.
Choosing a therapy that is safe, reliable, and supported by clinical data matters because the outcomes on the other side of that decision belong to a real person. That's why Thrombolex invests in the clinical programs, resources, and a Family of Endovascular Devices that give the interventional community the evidence and tools they need to deliver better outcomes for every patient.
Thrombolex.com #Thrombolex ... See MoreSee Less
0 CommentsComment on Facebook