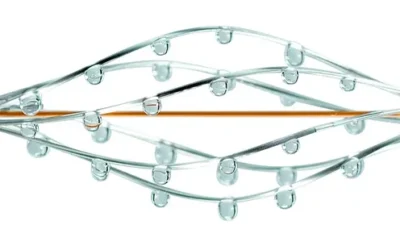
New Britain, PA, May 4, 2026 — Thrombolex, Inc., a medical device company dedicated to advancing minimally invasive endovascular treatment of venous thromboembolism (VTE), today announced the enrollment of the 100th patient in the RAPID-PE clinical study. This...
News
Our Latest News
Thrombolex Secures $50M Series A Financing to Advance Endovascular Treatment for Pulmonary Embolism and Venous Thrombus
New Britain, PA, March 16, 2026 – Thrombolex, Inc., a commercial-stage medical device company advancing a differentiated pharmaco-mechanical lysis (PML) platform for the treatment of pulmonary embolism (PE) and other thromboembolic diseases, announced the closing of a...
Interim Data from the RAPID-PE Study Presented at TCT 2025
New Britain, PA - (October 28th, 2025) - Thrombolex, Inc. presented the prespecified interim analysis from the first 50 patients in RAPID-PE, a prospective, multicenter U.S. study evaluating On-The-Table (OTT) pharmaco-mechanical lysis (PML) without any post procedure...
RESCUE-II Study results published in JACC: Advances
New Britain, PA – (June 25, 2025) – Thrombolex, Inc., a medical device manufacturer committed to advancing solutions for the treatment of arterial and venous thromboembolic diseases, announced the publication of the results from the RESCUE-II Study in JACC: Advances....
Rescue II Results
RESCUE II Trial Demonstrates the Feasibility of On-The-Table Pharmacomechanical Lysis without Post-Procedure Infusion in Treating Acute Pulmonary Embolism NEW BRITAIN, PA (October 28, 2024) — Christian Bichard, MD presented the results of the RESCUE-II Trial during...
RAPID-PE Study News
Thrombolex Announces the First Two Patients enrolled in the RAPID-PE Clinical Study evaluating an Advanced On-The-Table Protocol for the Treatment of Pulmonary Embolism NEW BRITAIN, PA (October 23, 2024) — Thrombolex, Inc. announced the enrollment of the first two...
Thrombolex and Aidoc Announce Strategic Partnership to Advance Breakthrough Pulmonary Embolism Treatment
Proactive collaboration leverages Aidoc's AI technology to accelerate RAPID-PE clinical trial enrollment for Thrombolex's "single session" treatment protocol. NEW BRITAIN, PA, July 30, 2024 – Thrombolex, Inc., an innovator in the development of advanced interventional...
First Patient Enrolled In Thrombolex’s RESCUE II Study — On-Table Pharmacomechanical Lysis Without Post-Procedural Infusion
NEW BRITAIN, PA (January 22, 2024) Thrombolex, Inc. announced the enrollment of the first patient in the RESCUE II study using the BASHIR™ Endovascular Catheter for the treatment of acute intermediate-risk pulmonary embolism by Drs. Riyaz Bashir and Vladimir Lakhter,...
Thrombolex, Inc. Announces the New BASHIR™ .035″ Endovascular Catheters, Now Compatible with a 0.035″ Guidewire for the Treatment of Acute Pulmonary Embolism
NEW BRITAIN, PA (January 9, 2024) Thrombolex, Inc., a medical device company that develops innovative devices for the treatment of arterial and venous thromboembolic diseases, announces an expansion of the existing product line with the new BASHIR™ .035 and BASHIR™...
Significant New Insights from the NIH-Sponsored RESCUE Trial with the BASHIR™ Endovascular Catheter Published in JACC: Advances.
NEW BRITAIN, PA (October 24, 2023) Thrombolex, Inc., a medical device company focused on developing innovative products for the treatment of arterial and venous thromboembolic diseases, announced never-before-reported major reductions in obstruction in all of the...
Thrombolex Receives 510(k) Clearance for Treatment of Acute Pulmonary Embolism
NEW BRITAIN, PA – April 20, 2023 – Thrombolex, Inc. has received 510(k) clearance for use of the BASHIR™ and BASHIR™ S-B Endovascular Catheters for the treatment of acute pulmonary embolism (PE). This PE specific clearance was based upon the positive results from the...
Marked reduction in segmental and main pulmonary artery occlusions shown in NIH-funded RESCUE Trial
Marked reduction in segmental and main pulmonary artery occlusions shown in NIH-sponsored RESCUE trial with the BASHIR™ Endovascular Catheter in Patients with Intermediate Risk Acute Pulmonary Embolism NEW BRITAIN, PA (March 6, 2023). Robert A. Lookstein MD, Executive...