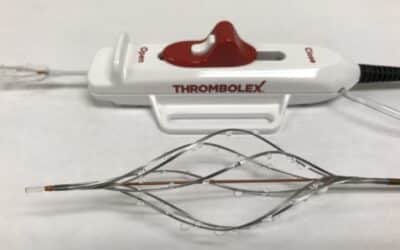
NEW BRITAIN, PA (December 15, 2022) Thrombolex, Inc. has been awarded a group purchasing agreement with Premier Inc. effective December 1, 2022. The new agreement allows Premier members, at their discretion, to take advantage of special pricing and terms...
News
Our Latest News
Thrombolex Announces Publication of RESCUE Trial Results in JACC, Cardiovascular Interventions
NEW BRITAIN, PA (December 8, 2022) Thrombolex, Inc., a medical device company focused on developing innovative products for the treatment of arterial and venous thromboembolic diseases, is proud to announce the publication of the results of the RESCUE trial. This...
Final RESCUE Trial Results Released At TCT 202
NEW BRITAIN, PA (September 17, 2022) Thrombolex, Inc. today presented the final results of its NIH - sponsored RESCUE trial during the Late Breaking Innovation Session at TCT2022 in Boston. This investigational device exemption (IDE) trial demonstrated that...
Michael Cerminaro to Succeed Marvin Woodall as CEO of Thrombolex
PRESS RELEASE - Woodall to Remain Executive Chairman of the Board of Directors New Britain, PA (July 15, 2022) Thrombolex today announced that its Board of Directors has named Michael Cerminaro Chief Executive Officer (CEO) of Thrombolex effective July 13, 2022, the...
BASHIR™ Endovascular Catheter Shows Marked Reduction in RV/LV Ratio and Thrombus
BASHIR™ ENDOVASCULAR CATHETER SHOWS MARKED REDUCTION IN RV/LV RATIO AND THROMBUS BURDEN IN PATIENTS WITH ACUTE INTERMEDIATE RISK PE: INTERIM RESULTS OF NIH-FUNDED RESCUE TRIAL New Britain, PA, October 5, 2021 THROMBOLEX, INC. today announced the results of the RESCUE...
Thrombolex, inc. Attendance at Viva & Veins in San Diego
THROMBOLEX, INC. will be attending the VEINS Conference (Booth T8) and VIVA Physicians Conference (Booth #502) in Las Vegas, NV October 3-7. There will be live demonstrations and meetings with the Executive Team about the family of BASHIR™ Endovascular Catheters. The...
How One Smart MedTech Company is Leveraging Innovation to Save the Lives and Limbs of VTE Patients
Medical device start-up companies must focus on innovation, regulatory compliance, efficient processes, and prudent financial management in order to survive. Smart MedTech companies prioritize their funding to only support innovation which will result in new products...
A Paradigm Shift in the Treatment of Venous Thromboembolic Disease
PE is the most common preventable cause of death among hospital patients in the United States. VTE is a leading cause of death and disability worldwide and has the third-highest mortality rate out of any cardiovascular disease, behind myocardial infarctions and...
Benefits of Achieving Robust Clot Resolution in VTE Patients…
Benefits of Achieving Robust Clot Resolution in VTE Patients While Balancing Lower Risk of Adverse Events By Michael J. Cerminaro , Brian G. Firth How Advanced Catheter-Directed Therapies May Help Improve Outcomes While Reducing the Incidence of Post-PE Syndrome...
First patient enrolled in RESCUE trial of Thrombolex’s Bashir catheter
Thrombolex has announced that it has enrolled the first patient in its pivotal RESCUE trial for the treatment of patients with acute submassive pulmonary embolism (PE) using the Bashir endovascular catheter, under an Investigational Device Exemption (IDE) from the US...
The Search for the Holy Grail of New PE Devices
BY MAHIR ELDER, MD, FACC, FSCAI, AND TERRY BOWERS, MD, FACC, FSCAI A review of interventional thrombolytic and catheter-based therapies for PE treatment. Pulmonary embolism (PE) is a growing field in endovascular intervention with a robust ongoing search for the...
Benefits of Achieving Robust Clot Resolution In VTE Patients Article
The Benefits of Achieving Robust Clot Resolution In VTE Patients While Balancing Lower Risk of Adverse Events – Brian Tweddale, MD, Doylestown Hospital The majority of VTE patients have traditionally been treated with anticoagulation alone. However, up to 50% of...