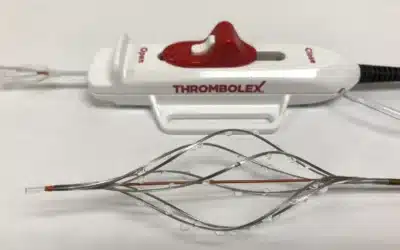
THROMBOLEX™ Inc. Receives $3 million Small Business Innovation Research (SBIR) grant from the NIH to fund the RESCUE trial. Thrombolex™ Inc. has announced that it was awarded a $3 million Small Business Innovation Research (SBIR) grant from the National Heart, Lung...
News
Our Latest News
First-in-human results for Bashir Endovascular Catheter meet safety and feasibility endpoints
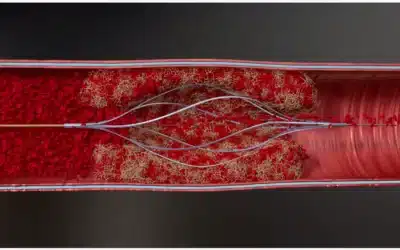
Thrombolex has announced that results of its First-In-Human (FIH) trial confirm the early safety and feasibility of using the Bashir Endovascular Catheter for pharmacomechanical catheter-directed thrombolysis (CDT), in patients with acute pulmonary embolism (PE). The...
Thrombolex is honored by receiving Temple University’s Prestigious President’s Spin-Out Award
Temple University President, Richard Englert, and Vice President for Research, Michele Masucci, gathered colleagues to honor the University’s top inventors, and distinguished researchers at the third annual President’s Innovation Award Gala. Fueled by more than $279...
Thrombolex™ Awarded $1 Million State Grant for Revolutionary Medical Device Research
Thrombolex Inc. executives joined State Senators Pat Browne (R-16 District) and Steve Santarsiero (D-10 District) to announce a $1 million state grant awarded to Thrombolex. The state funding will support clinical trials for Thrombolex’s BASHIR family of catheters,...
Thrombolex Nabs 510(k) Clearance for Catheter System
The FDA has granted Thrombolex 510(k) clearance for its Bashir Endovascular Catheter, a device used to manage the infusion of fluids. The device is used by physicians to control the infusion of specific fluids, such as thrombolytics, into a patient’s peripheral...
Thrombolex announces 510(k) clearance of Bashir catheter systems for thromboembolic disorders
Thrombolex has announced that the US Food and Drug Administration (FDA) has cleared the Bashir Endovascular Catheter (BEC) for the controlled and selective infusion of physician-specified fluids, including thrombolytics, into the peripheral vasculature, and the Bashir...
Thrombolex Receives FDA Clearance for Bashir Endovascular Catheters
May 13, 2019—Thrombolex, Inc. announced FDA 510(k) clearance for the Bashir endovascular catheter (BEC) and the Bashir N-X endovascular catheter (BEC N-X). The BEC and BEC N-X are currently only available in the United States. Plans for distribution outside the United...